By Nidhi Parekh, The Shared Microscope
Volunteering to receive an experimental COVID-19 vaccine is by no way a small feat. It can be difficult to delve into the unknown. Yet, around the world, thousands have dedicated their time to participating in human clinical trials testing the safety and efficacy of these vaccines.
Volunteering to receive an experimental COVID-19 vaccine is by no way a small feat. Yet, globally, thousands have dedicated their time to participating in human clinical trials of COVID-19 vaccines.
The Oxford/AstraZeneca clinical trials are being conducted in South Africa, Brazil, and the UK. The vaccine is currently in simultaneous Phase 2 and Phase 3 trials in England, and Phase 3 trials in Brazil. The trials underway help ensure the vaccine’s safety and efficacy.
We expect to hear about the results of these clinical trials — just one of several COVID-19 vaccines in the race to control the novel coronavirus pandemic — by Fall 2020. Anticipating the success of these clinical trials, and given the urgent need for a COVID-19 vaccine, Oxford and AstraZeneca have already started manufacturing the vaccine to make up to 400 million doses by late 2020.
In this article, we had the opportunity to interview Mr. Zan Markan, an IT professional living in the UK. Mr. Markan is a participant in the clinical trials for the COVID-19 vaccine being developed by Oxford. As part of the study, he has a 50% chance of having received the experimental vaccine. Half of the clinical trial participants receive the Oxford vaccine, and the other half receive a meningococcal vaccine that is already on the market as a control. This experimental design using a vaccine as a control ensures that all participants will experience similar post-injection side effects, such as pain at the injection site, fever, etc., which will help participants be less able to determine whether they got the COVID-19 vaccine or the control vaccine.
So far, Mr. Markan has reported feeling completely healthy.
Mr. Markan was recruited for the trial at the end of June 2020. His first public statement about the clinical trial was over his Twitter account on July 04, 2020, where he described the science behind the vaccine as “absolutely kick ass.”

The Oxford/AstraZeneca COVID-19 Vaccine And How It Works
The Oxford/AstraZeneca vaccine is developed by the Jenner Lab in Oxford. The lab originally worked on inventing a vaccine for a related coronavirus called MERS-CoV which causes Middle East Respiratory Syndrome. However, with the COVID-19 pandemic, the Jenner Lab decided to shift their efforts.
The Oxford vaccine is effectively made by the merging together of two viruses – the ChAdOx1 virus, which causes cold-like symptoms in Chimpanzees, and the SARS-CoV-2 virus, which causes COVID-19. From the outside, the active ingredient of the vaccine looks like a capsule – this capsule is made of the ChAdOx1 virus. Inside this ChAdOx1 capsule, is the genetic information for the SARS-CoV-2 virus’ spike protein only. The viruses contained in the vaccine are engineered so that they are unable to cause disease. In fact, they teach our body how to prevent future COVID-19 infections. To learn more about this, please check out our full post on the Oxford/AstraZeneca vaccine.
Doing It “For Science”
When asked about his decision in partaking in the Oxford/AstraZeneca Study, Mr. Markan’s reasoning was simple: in his view, there was a “low risk and a potential reward.” He also thought of it as “something [he] can do to help in the efforts against COVID-19.” He first heard about the Oxford study on the news, and knew that the researchers were looking for volunteers. This is when he first enrolled as a participant. A couple days later, the Oxford researchers contacted him for more information and also for an enrollment meeting.
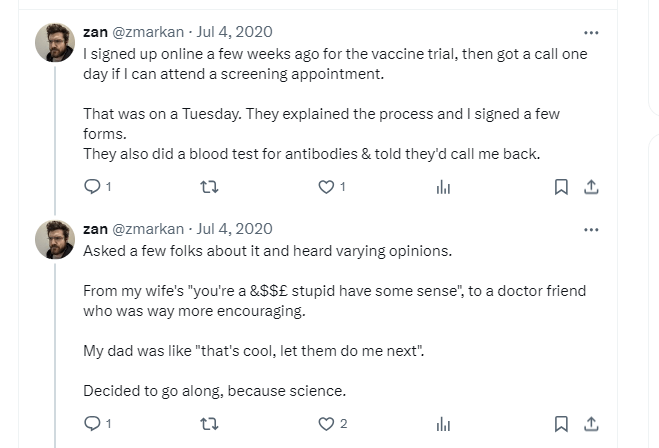
Before he could be formally enrolled in the study, the researchers had to make sure he was healthy to begin with – so he went to the hospital for his initial screening appointment. There, Mr. Markan participated in a blood test to make sure that he had no existing antibodies against the SARS-CoV-2 virus. Had he been infected with the SARS-CoV-2 virus prior to the study, he would not be allowed to partake in it, because his body would in theory already have the antibodies needed to fight the COVID-19 infection. The vaccination would be futile in this case. Luckily, as he tweeted, his antibody test results showed he had no COVID-19-specific antibodies, which meant he could enroll in the study.

Mr. Markan admits to doubting his decision, as can be seen from his tweet above. However, he went on to become a candidate in the study, after talking to a doctor friend who assured him that the vaccine had already been through many rounds of rigorous clinical trials.
“A Risk I’m Willing To Take”
Mr. Markan is a self-described “techie.” “You know more about the science behind the vaccine than I do,” he explained in this interview. However, he was motivated to participate in the clinical trials due to the low risk involved in the study. He was able to conclude that the risk to his life was “minimal,” albeit unknown. Different friends and family had varying opinions.
After the various responses he got from friends and family, and his first meeting with the researchers, Mr. Markan told us that being in the trial was a “risk [he was] willing to take.”
Participating in the Oxford/AstraZeneca COVID-19 vaccine trials was a risk Zan Markan was willing to take to benefit the greater good.
The Consultations
In a single initial consultation, researchers explained the risks, went through paperwork, and underwent laboratory testing. During this consultation, the researchers drew some blood to ensure he had no antibodies against the disease. Once it was confirmed that he did not have antibodies for SARS-CoV-2, the virus that causes COVID-19, he had a subsequent consultation to confirm he was still willing to continue with the trial — and obtain the vaccine. The researchers discussed with him some potential risks and side effects post-vaccination. This included risk for a severe allergic reaction.
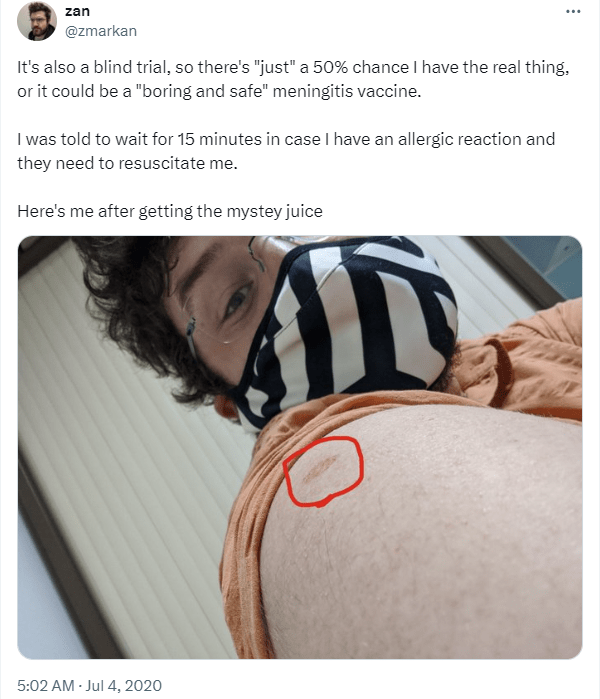
To ensure that he wouldn’t have a severe allergic reaction post-vaccination, Mr. Markan was kept under observation for about 15 mins in the case of such an adverse event. He was also told of other side effects, including some pain in the area of his arm where he was injected, a fever, and cold-like symptoms. It was explained to Mr. Markan that, should anything happen, he would have direct contact with the research team, and would also be given care in the hospital; however, so far, the only symptom he has reported so far is some mild pain around the site of injection.

Throughout all of his consultations and his enrollment meeting, Mr. Markan was encouraged to ask questions. If he was agreeable to all the potential side effects (i.e., gave consent), only then would he receive a vaccine (whether placebo or the Oxford vaccine). The researchers also informed Mr. Markan that he could opt out of the study at any time.
Participating in a COVID-19 Vaccine Trial Is As Easy As 1, 2, 3
To get a well-rounded picture of Mr. Markan’s experience as a study participant, we wanted to know more about the time he spends on it. After receiving the jab, Mr. Markan was “sent home with four sets of COVID-19 tests.”
Mr. Markan described the whole testing process as “super easy.” He added that “every Thursday, [he] gets a text asking to do a swab test per the instructions and put the vial in the box. Then you go online, register your barcode with your profile, and get results – you send this over to [the researchers].” He mentioned that the throat and nasal swabs are not difficult to do, although they could be uncomfortable for someone with a strong gag reflex. He does not have this problem. His results are marked for expedited shipping (to get to the testing lab as soon as possible) and also have a stamp that helps them get to the lab quickly. The lab work, once completed and packaged up into the dedicated mailing box, is dropped off into a Priority Postbox, like the one shown below.

Once received by the laboratory, these results get processed every evening, and the results are sent to him via text message and email the next day. The entire process of getting his swab samples and sending them off takes him about 30 minutes, including walking to the post box and sending off his samples.
After the four COVID-19 test kits are used, he mentioned that he “will get invited to do another set of blood work that will take an hour or so – that’s it. They will then give [him] another four test kits. After eight weeks, they may extend this trial for up to one year. If the vaccine does not work, they will know that, and stop the trial. If the trial proves to work, [he] will keep swabbing until they have enough data to proceed or stop the trial.”
Going Out and About as a COVID-19 Vaccine Clinical Trial Participant
Since Mr. Markan has possibly been injected with a modified version of the SARS-CoV-2 virus, we were interested to know more about whether he is allowed to go out during the pandemic. He responded by saying that “the idea is to keep living life as you would have. As someone who stares at the screen for the whole day, I’m not particularly in a vulnerable group to get COVID-19, but they wanted a wide sample of the whole populace. There is no doing stupid things. They just reopened our pubs in the UK so a subset of people will be doing stupid things, but I try the sensible thing and avoid exposure. It is a pretty serious disease.”
He also reminded us that he is in a trial and does not know whether he received the COVID-19 vaccine or a placebo vaccine for meningitis. “The placebo vaccine causes very similar symptoms to the COVID-19 vaccine – flu-like symptoms, arm ache, fever … if you get a fever, cause they don’t know whether it’s from the vaccine or actual COVID-19 or another reason that people have a fever – it could be COVID – then you are instructed to self-isolate with the whole household. If the fever kind of subsides within 72 hours – you can start going along as you go.”
Participants in the UK Oxford/AstraZeneca COVID-19 clinical trials have a 50% chance of receiving either the COVID-19 vaccine or a placebo (a meningitis vaccine).
Mr. Markan is hopeful that the vaccine will work, and help us take the final steps towards ending this pandemic.
Conclusion
Mr. Markan’s participation in this study is an act of altruism of the highest form; he is a very important statistic worldwide. How his body reacts in this study will be used as a data point for a number of individuals worldwide. It is important to note that Mr. Markan is not getting compensated for his participation in this study. Should anything deviate from the plan, he would be taken care of by the NHS, as his work-based health insurance company has refused to cover any COVID-19 related illnesses.
Mr. Markan added that he was partaking in this trial “for science.” During our interview, he mentioned that enrolling in the study was “a risk [he was] willing to take” and that he “was not afraid of vaccines.” He looks forward to “trying this test thing and contributing as a data point to something that can really make a difference in the long run.”
Nidhi Parekh blogs about science at The Shared Microscope. Check out her Twitter, Facebook, or Instagram for informative illustrations of different biological concepts.
.
Reblogged this on The Shared Microscope.