Updated February 12, 2022
By Nidhi Parekh of The Shared Microscope and Sheeva Azma of Fancy Comma, LLC
The Moderna vaccine is an mRNA-based vaccine which helps the human body develop immunity to SARS-CoV-2, the virus that causes COVID-19. In this post, learn about the Moderna vaccine for COVID-19 and how this mRNA vaccine works. Lea este artículo en español.
How does the #Moderna COVID-19 vaccine work? Read this post to find out. $MRNA
Tweet
Check out other posts in our COVID-19 vaccine series:
[Moderna | Oxford/AstraZeneca | Pfizer/BioNTech | Johnson & Johnson | Novavax | Sinovac]
Navigate to a particular section of this post:
- The race to a COVID-19 vaccine
- How do vaccines work?
- What are vaccines normally made of?
- The Moderna vaccine development process took only 42 days
- How does the Moderna vaccine work?
- It gives our bodies the instructions to manufacture a small piece of the novel coronavirus, called a spike protein, which is responsible for a COVID-19 infection.
- Once our cells produce the spike proteins, our immune system neutralizes them, and we develop immunity to COVID-19.
- Nucleic acid vaccines
- Could COVID-19 bring mRNA vaccines to the limelight?
The Race to a COVID-19 Vaccine
In the midst of the pandemic, the race to a COVID-19 vaccine is very competitive. Several companies have emerged as leaders in COVID-19 clinical trials, and the first to emerge successful from the clinical trials process could end the COVID-19 pandemic as we know it.
A federal U.S. initiative called Operation Warp Speed is supporting the parallel development of several vaccines from major pharma companies via upwards of $13 billion in investment from the U.S. Department of Health and Human Services (HHS):
- Moderna’s mRNA-1273
- AstraZeneca and Oxford University’s AZD1222
- A promising candidate vaccine from Johnson & Johnson
- A Merck vaccine based on their successful Ebola vaccine
- Novavax’s NVX-CoV2373
Moderna’s mRNA-1273 is arguably one of the most successful COVID-19 vaccine trials at the moment. Check out its rapid development timeline on the Moderna website. Moderna went from mRNA sequence selection stage to clinical testing in humans in only 42 days!
After promising results on its FDA Phase 2 clinical trials, the Cambridge, MA-based company begins its Phase 3 clinical trials in July, and will be the first company to do so. Phase 3 trials are meant to examine the overall risks and benefits of the drug in humans. If this stage of the trial is successful, the vaccine could be the first in the world to be approved by the FDA to prevent COVID-19 infection and help end the pandemic.
How Do Vaccines Work?
Think of your immune system as your body’s natural antivirus software. Vaccines are a way to update your body’s antivirus software – literally!
Vaccines can be used for viruses such as COVID-19, as well as other pathogens — unwanted foreign invaders — such as bacteria and microorganisms. Our bodies have the ability to develop novel responses against them.
Vaccines work by encouraging the body to create special molecules called antibodies. The purpose of antibodies is to defend our bodies against infection. Antibodies directly interact with the invader or antigen to neutralize and destroy it. By doing so, antibodies help keep us healthy.

Antibodies are Y-shaped proteins important in our body’s immune response. Source: The Shared Microscope on Instagram.
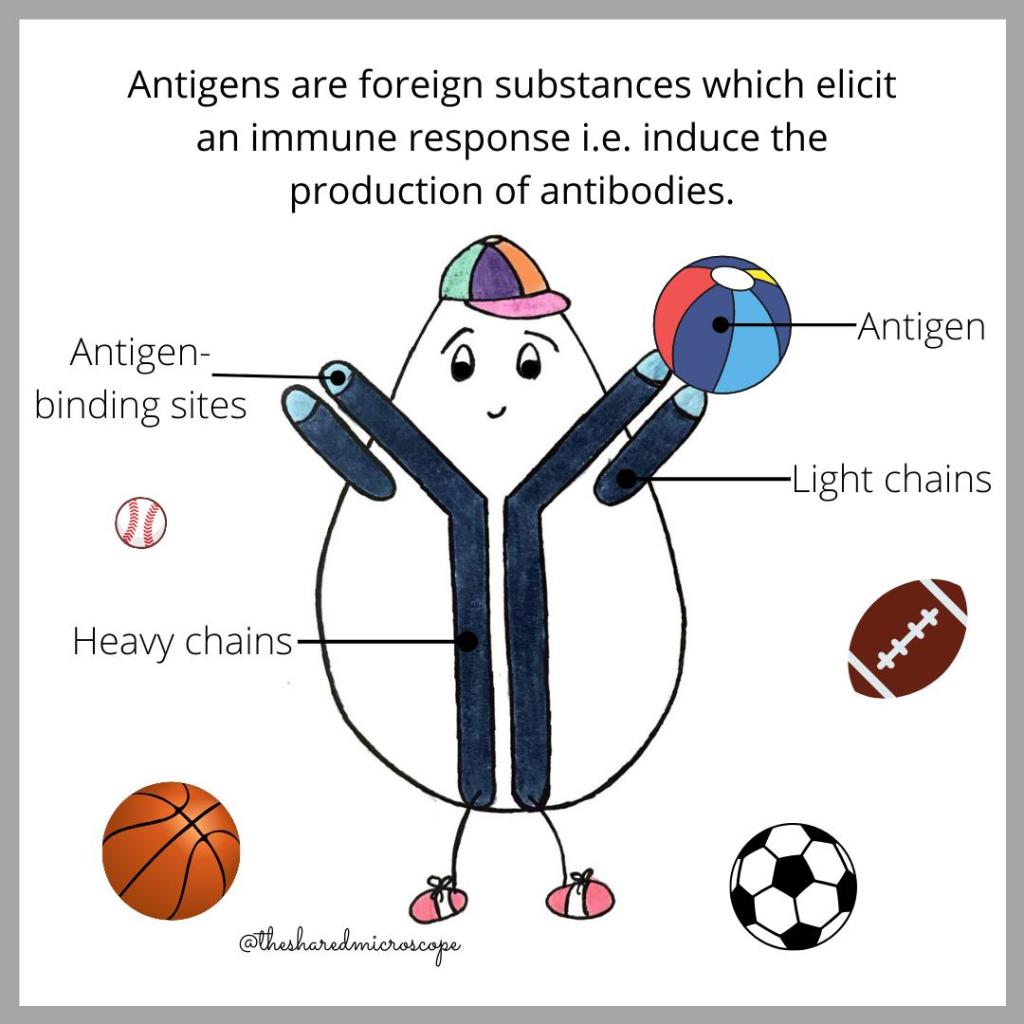
Antibodies help us fight off pathogens like SARS-CoV-2, the virus that causes COVID-19. Source: The Shared Microscope on Instagram.
What Are Vaccines Normally Made Of?
While all vaccines work the same way — by encouraging the production of antibodies to fend off future infections — Moderna’s mRNA-1273 is different. This is because Moderna’s vaccine uses different material — mRNA of the virus, rather than a weakened or inactivated version of the virus itself — to follow through to the same result. For this reason, it was much faster to develop.
There are several types of conventional vaccines, including live attenuated vaccines, inactivated vaccines, subunit vaccines, and toxoid vaccines. The goal of these vaccines is to introduce a small amount of the virus into the body so that our immune system can attack the virus without us getting sick, so that we develop immunity to the virus.
Moderna’s Vaccine Development Process Took Only 42 Days
Unfortunately, the process of developing these conventional vaccines can be very time-consuming, taking anywhere from 10 to 15 years, and expensive, costing anywhere from $200 million to $1 billion. Check out the illustration below to learn more about the different stages of a vaccine clinical trial.

Vaccine development has many different phases and can normally take over a decade. There is a global effort to accelerate vaccine development efforts in hopes of a swift end to the COVID-19 pandemic. Source: The Shared Microscope.
10 years is a long time, especially in the context of the ongoing pandemic as we work daily to mitigate the spread of COVID-19 through wearing masks, social distancing, and other measures that have impacted our economy.
Thanks to advances in both computing and genetic sequencing, Moderna’s researchers were able to take the vaccine from an idea in the lab to clinical testing in 42 days. In this time, Moderna was able to both identify an area of the spike protein of interest and select a region of the spike protein mRNA to be used for the vaccine.
The @Moderna_tx mRNA #COVID19 #vaccine took 42 days to go from sequence selection to clinical trials. Typical vaccines take much longer — 10+ years — to get to that point. #scicomm @TheSharedScope
Tweet
How Does Moderna’s COVID-19 Vaccine Work?
In short, the vaccine helps us develop immunity to a small, yet very important, part of the SARS-CoV-2 virus: the spike protein found on the virus’s surface.
Let’s refer to the iconic image of SARS-CoV-2 below to illustrate the SARS-COV-2 spike proteins and why they’re so important in COVID-19 infection. The red spikes on the novel coronavirus molecule are the spike proteins. These spiky proteins allow SARS-CoV-2 RNA to gain entry to our body’s cells. This way, the novel coronavirus enters cells, instructing them to make more of the novel coronavirus. This process is what causes COVID-19 infection.
Moderna’s solution? Help the body develop immunity to the spike proteins. That way, if spike proteins are found in our bodies, they will be broken down by the immune system, and there will be no way for SARS-CoV-2 to enter our cells. Without a way in to our cellular machinery, SARS-CoV-2 cannot replicate, and a COVID-19 infection can not take hold.

SARS-CoV-2, the virus that causes COVID-19. Spike proteins are in red.
The Moderna mRNA Vaccine Contains Instructions for Our Bodies to Create SARS-CoV-2 Spike Proteins
Moderna’s vaccine is unique as it is based on messenger RNA (mRNA), rather than the virus itself. Messenger RNA are cellular “messages” which instruct the body to manufacture proteins.
The mRNA-1273 vaccine contains the mRNA needed to create the spike protein found on the surface of the SARS-CoV-2 virus — the virus that causes COVID-19. In other words, the vaccine contains the genetic code of the spike protein. The spike protein is crucial to SARS-CoV-2, as it helps the virus enter our cells and take over our cellular machinery to produce and assemble more viruses and viral particles.
Once the vaccine is administered, the mRNA instructs our body to produce only the spike proteins. Our bodies produce the spike protein without becoming infected by the virus. Even though these proteins are produced in our bodies, our immune system recognizes them as invaders, or antigens. This is because the spike protein is not normally found in a healthy individual.
With the help of this vaccine, the body is able to learn to identify the spike protein as an invader, and make antibodies to neutralize and then destroy the spike proteins. This is essential to prevent further infection by SARS-CoV-2.
So the answer to the the pandemic is antibody production — that is, antibodies produced in response to the SARS-CoV-2 virus.
The idea here is that developing immunity against the spike protein could prevent infection to SARS-CoV-2 even if we are exposed to it, because our body will know how to break down the spike proteins. Without the spike proteins, the virus cannot get into our cells and cause COVID-19 — making us immune to the virus.
Curious about innovative vaccine candidates to defeat the novel #coronavirus? The Moderna vaccine contains the mRNA code for the spike protein found on the surface of the #SARS-CoV-2 virus — the virus that causes #COVID19. @TheSharedScope
Tweet
The Moderna Vaccine Teaches Our Immune System How to Break Down SARS-CoV-2 Spike Proteins
The Moderna vaccine helps our immune system come up with a “game plan” to break down the novel coronavirus’s spike proteins. Without spike proteins, the SARS-CoV-2 virus cannot enter our cells.
Check out this informative series of infographics from The Shared Microscope on how the Moderna vaccine works:
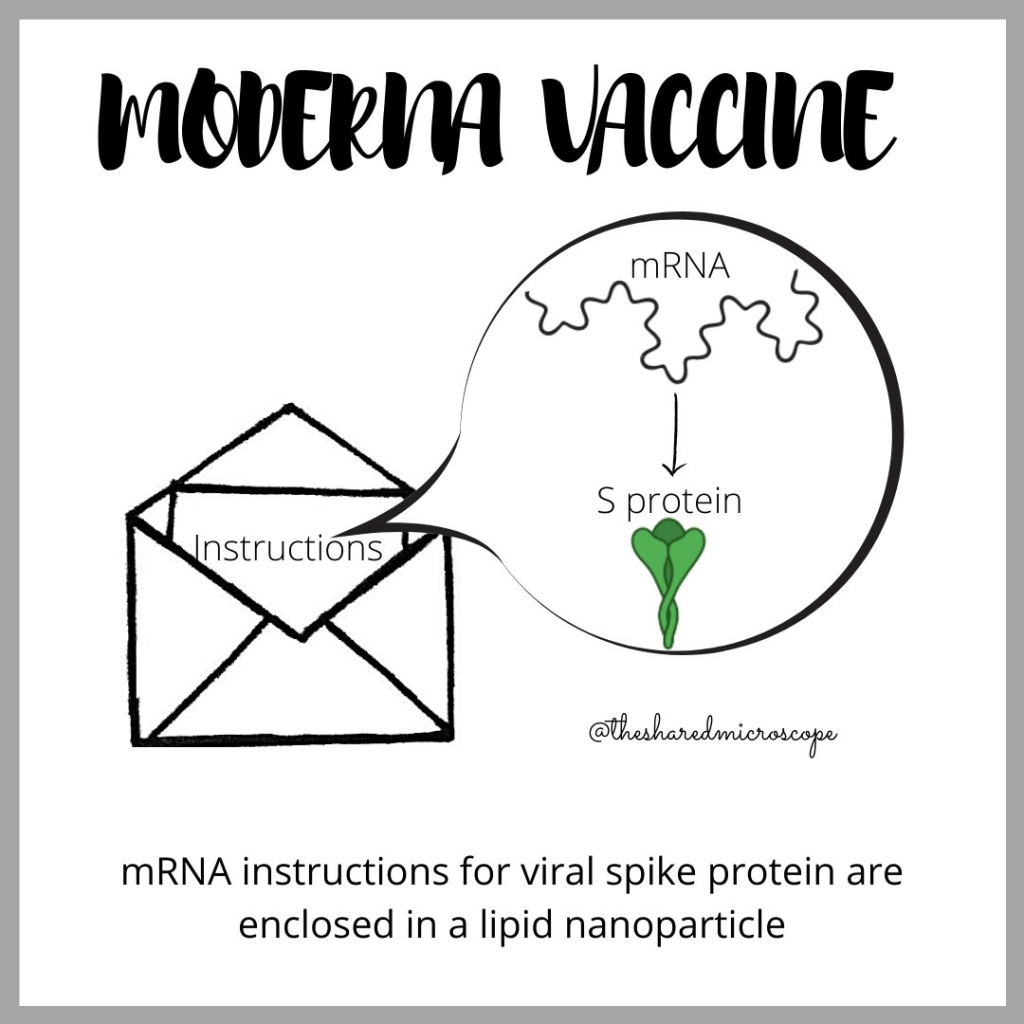
The vaccine contains mRNA that tells our cells how to make only the spike proteins of SARS-CoV-2. Source: The Shared Microscope on Instagram.
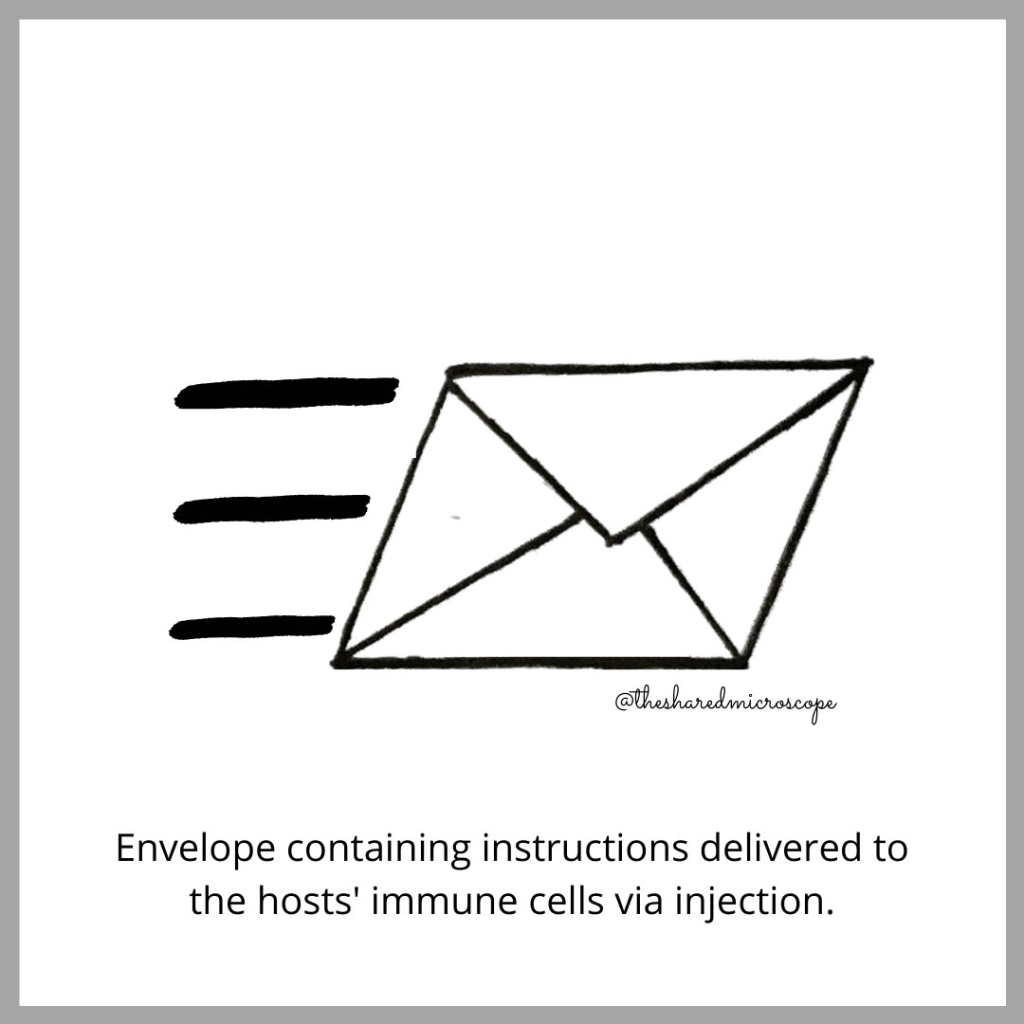
The vaccine is injected into our bodies, delivering the instructions for making spike proteins directly to our cells. Source: The Shared Microscope on Instagram.

The body reads the instructions and produces only the spike proteins of SARS-CoV-2, the virus that causes COVID-19. Source: The Shared Microscope on Instagram.

The body develops antibodies to neutralize the spike proteins. The next time a SARS-CoV-2 virus enters our body, the antibodies ensure that the spike protein can be neutralized, so the virus can not gain access to our cells. Source: The Shared Microscope on Instagram.
For more information, check out this video from Moderna which offers a detailed explanation of how mRNA-1273 works:
In summary, the Moderna vaccine serves as a carrier pigeon, delivering our cells the necessary instructions to build the SARS-CoV-2 spike protein, so that we can develop immunity to it and avoid being infected with COVID-19.
@Moderna_tx’s vaccine acts as a carrier pigeon, delivering our cells the needed instructions to build the SARS-CoV-2 spike protein, so we can develop immunity to the novel #coronavirus and avoid #COVID19 infection. @TheSharedScope
Tweet
The New and Rapidly Growing Field of Nucleic Acid Vaccines
At this early stage of testing, Moderna’s vaccine looks promising, but nobody is sure whether this vaccine will be the one to end the pandemic. However, the use of mRNA in a vaccine is a first, not only for Moderna but for the world.
Moderna’s mRNA-1273 is an example of a vaccine that uses nucleic acids such as DNA or mRNA. Nucleic acid vaccines are faster to develop than traditional vaccines because of their reliance on computational modeling of vaccine interactions, which cuts down on the scientific experimentation needed. For this reason, they are cheaper to develop compared to conventional vaccines, and they are cheaper to administer to patients.
Moderna, who has eight mRNA vaccines in the pipeline besides mRNA-1273, discusses several advantages of mRNA vaccines on their website, namely:
- Ability to Mimic Natural Infections: Nucleic acid vaccines, such as Moderna’s mRNA vaccine, can mimic many aspects of natural infections, and could be more effective than conventional vaccines because they evoke stronger immune responses.
- Ability to Include Many Viral mRNA Particles in a Single Vaccine: Many different mRNA corresponding to different viral proteins could be included in a single vaccine. This could be useful to provide many different viral mRNA segments in a single dose, e.g., to reduce the total number of immunizations a person may need, or create antibodies for different aspects of the same pathogen.
- Extremely Rapid Vaccine Development: Computational modeling and design of mRNA antigens is much, much faster in silico (using computer modeling) than in the laboratory. High-throughput screenings can be conducted on a very rapid timescale in the computer lab (e.g., examining the best candidates of a virus’s mRNA to be used in a vaccine). Then, the most theoretically promising candidates can then be tested at the scientific bench.
- Saving Time Saves Money: Cost savings are gained from the efficiency and speed by which such vaccines can be developed, owing to shared manufacturing processes and infrastructure.
Could COVID-19 Bring mRNA Vaccines into the Limelight?
If approved, mRNA-1273 would be the first mRNA vaccine on the market anywhere in the world, and possibly the first COVID-19 vaccine, to boot. To expedite the race to a COVID-19 vaccine, Moderna has recently pledged that it will not enforce its COVID-19-related patents, which means that its technology could potentially be used in future vaccines as well.
If approved by the FDA, Moderna’s mRNA-1273 would be the first #mRNA #vaccine on the market anywhere in the world, and possibly the first #COVID19 vaccine, to boot. @TheSharedScope
Tweet
Will Moderna be the victor in the race towards a COVID-19 vaccine? Only time will tell. For now, though, the world is incredibly grateful for all of the institutions out there working towards a vaccine to end the global pandemic and help us move forward and get back to a “new normal” in which it is once again safe to go about our daily lives.
Update: On December 18, 2020, the Moderna vaccine became the second COVID-19 vaccine, and the first vaccine part of Operation Warp Speed, to obtain FDA approval via an Emergency Use Authorization. On January 31, 2022, the vaccine obtained full FDA approval for persons aged 18 and older, commercialized under the trade name “Spikevax.”
This post is a collaboration with Nidhi Parekh of The Shared Microscope. Check out their Twitter, hop over to their Facebook, or visit their Instagram for informative, effective illustrations of concepts in biology.
25 thoughts on “How Does Moderna’s mRNA Vaccine for COVID-19 Work?”