Updated August 26, 2021
By Nidhi Parekh of The Shared Microscope
Learn more about the Pfizer/BioNTech COVID-19 vaccine. $PFE $BNTX
Tweet
The Pfizer and BioNTech vaccine was a “dark horse” in the COVID-19 vaccine race, but the first to obtain approval from the U.S. Food and Drug Administration (FDA). Read on to learn about how the Pfizer/BioNTech vaccine works, and the clinical trials process more generally. Please note that while this vaccine is commonly referred to as the Pfizer/BioNTech or Pfizer vaccine (which we will use interchangeably in this blog post), it is also being developed in collaboration with Chinese drug maker Fosun Pharma.
Update: This vaccine obtained FDA approval for use in adults via Emergency Use Authorization on December 11, 2020, making it the first commercially available COVID-19 vaccine. On May 10, 2021, the vaccine became authorized for emergency use in adolescents aged 12-15 as well. On August 23, 2021, the vaccine gained full approval from the FDA. Obtaining FDA approval gives Pfizer/BioNTech the ability to market their vaccine (called Comirnaty) directly to consumers in the United States.
Check out other posts in our COVID-19 vaccine series:
[Moderna | Oxford/AstraZeneca | Pfizer/BioNTech | Johnson & Johnson | Novavax | Sinovac]
Table of Contents:
- The Clinical Trials Process for Vaccines
- Pfizer/BioNTech’s Clinical Trials: “Warp Speed”
- The Pfizer/BioNTech Vaccine Is an mRNA Vaccine
- Pfizer and BioNTech’s mRNA Vaccine Is Called BNT162
- An Introduction to The BNT162b2 Vaccine
- The Pfizer/BioNTech vaccine relies on part of the novel coronavirus called the spike protein.
- What’s in the BNT162b2 Vaccine?
- How Does the Pfizer/BioNTech Vaccine Work?
- The Pfizer/BioNTech Vaccine’s Current Situation
The Clinical Trials Process for Vaccines
Vaccines have helped wipe out a number of deadly infections such as smallpox. As such, vaccines are one of the most important medical inventions in the world, but creating a vaccine can be a long and complex process. Vaccines need to go through a plethora of checks in clinical trials to ensure they are safe and effective – if vaccines don’t pass this test, they do not get FDA approval to be used or sold commercially.
The typical vaccine development process is long — both by design and out of necessity.
Tweet
The typical vaccine development process is long — both by design and out of necessity. It generally starts with in vitro and animal studies in the lab, followed by Phase I, II, and III trials in humans to demonstrate safety and efficacy. After completing Phase III trials, the vaccine has demonstrated that it is safe and effective and can obtain FDA approval. Even after FDA approval, ongoing safety and efficacy trials remain underway in Phase IV.

Vaccine development timeline, from preclinical laboratory research to clinical trials with human subjects. Illustration by Nidhi Parekh of The Shared Microscope.
Pfizer/BioNTech Clinical Trials: “Warp Speed”
Pfizer is collaborating with German company BioNTech to develop a COVID-19 vaccine in a timely manner. This vaccine, like those being developed by Moderna, Oxford University/AstraZeneca, Novavax, and Sinovac, is another promising candidate COVID-19 vaccine in the race against the pandemic.
The clinical trials for the Pfizer/BioNTech vaccine were entirely funded by Pfizer and it is not funded as part of Operation Warp Speed. However, the Pfizer/BioNTech vaccine was the first of the COVID-19 vaccines to obtain FDA approval via Emergency Use Authorization (EUA). While the Pfizer/BioNTech vaccine is not formally part of Operation Warp Speed, the U.S. government has already committed to purchasing millions of units of this vaccine.
In July 2020, the U.S. government offered Pfizer and BioNTech $1.95 billion to help the United States procure 100 million doses of the vaccine. As per this deal, the U.S. may acquire up to 500 million additional doses of the Pfizer/BioNTech vaccine. The two companies may also supply the European Union with 200 million doses as well, according to a recent press release.
Though the Pfizer/BioNTech vaccine was a latecomer to the COVID-19 vaccine race, development of this vaccine progressed at a breakneck speed, surpassing that of the vaccines in Operation Warp Speed. Clinical trials on the Pfizer/BioNTech vaccine started in July, when both Moderna and Oxford/AstraZeneca were in Phase 3 trials. However, the company’s CEO, Albert Bourla, was adamant that the vaccine would receive FDA approval by the end of 2020 — which it did on December 11, 2020.
The Pfizer/BioNTech Vaccine Is an mRNA Vaccine
Like the Moderna vaccine, the Pfizer/BioNTech vaccine uses messenger RNA or mRNA. It is an mRNA-based vaccine.
As we have explained in our previous post about Moderna’s mRNA-1273 vaccine, mRNA vaccines are developed by the most cutting-edge of technologies. mRNA vaccines contain small segments of mRNA developed for use as a vaccine to help the body establish immunity to a pathogen (such as the novel coronavirus, SARS-CoV-2). mRNA vaccines are a type of nucleic acid vaccine — a vaccine which uses nucleic acids such as mRNA or even DNA to elicit an immune response.
What Is an mRNA Vaccine?
Published scientific papers highlight that mRNA vaccines are a promising alternative to conventional vaccine approaches. Thanks to advances in technology that enable rapid genetic sequencing and analysis, mRNA vaccines can be rapidly developed. They are also comparatively cheaper to manufacture and can be safer to administer than conventional vaccines.
mRNA vaccines have several advantages in clinical trials and in distribution. They can be rapidly developed, are cheaper to manufacture, and may be safer to administer than conventional vaccines. $MRNA $BNTX
Tweet
An mRNA vaccine is made of mRNA — a messenger molecule commonly found in the cells of our body. Traditional vaccines, on the other hand, use parts of the pathogen (perhaps a protein as in the case of subunit vaccines like Novavax) or whole inactivated pathogens (as in the case of CoronaVac). These mRNA “messages” provide instructions to our bodies to create various proteins needed for survival.
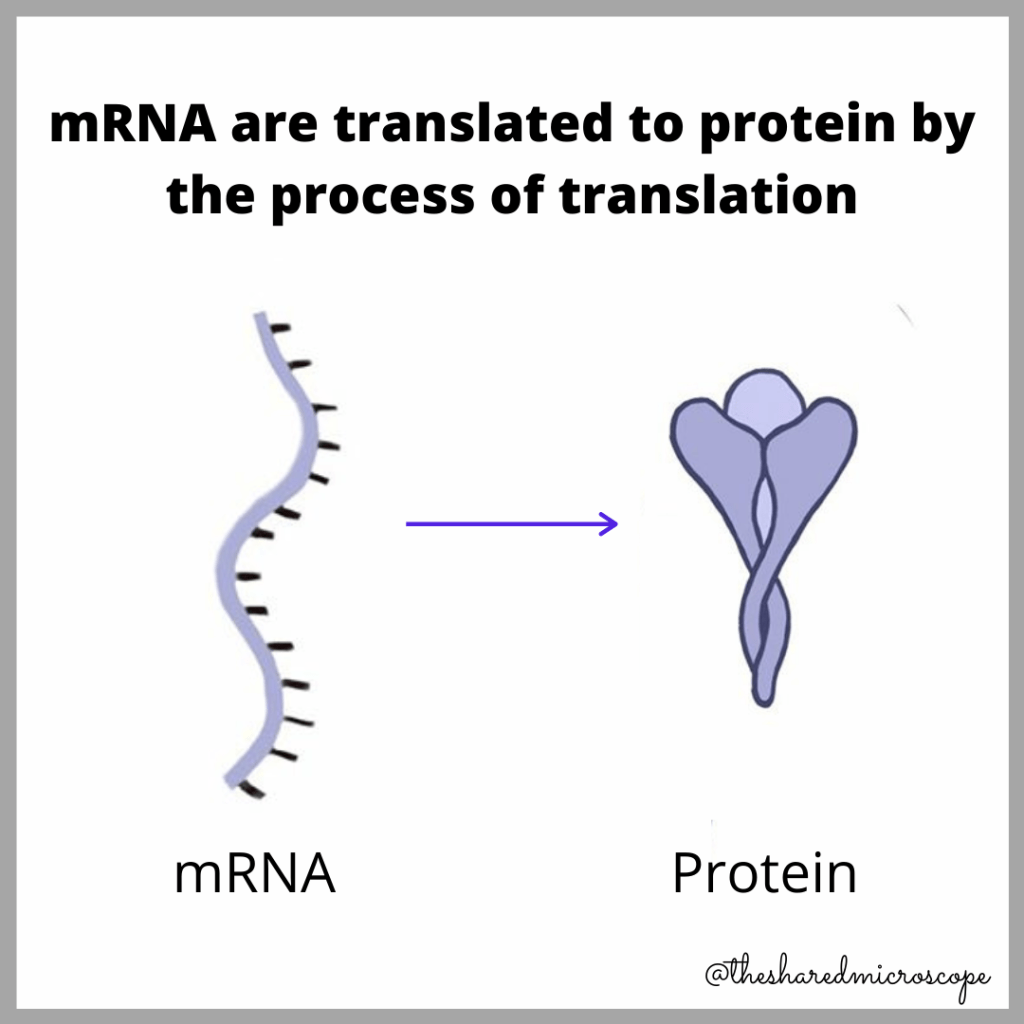
How Do mRNA Vaccines Work?
mRNA vaccines deliver mRNA to the body via injection. Once delivered to the body via a vaccine, the mRNA can tell the body’s cells to create the proteins needed to develop immunity to the antigen. In the COVID-19 vaccine race, the two most notable nucleic acid vaccines and, more specifically, mRNA-based vaccines, are the Pfizer/BioNTech vaccine and the Moderna vaccine.
The mRNA vaccines in development against COVID-19 carry the instructions vital for the production of spike proteins that are normally found on the surface of the novel coronavirus. These spike proteins are vital for infection. Neutralizing the spike proteins via the body’s natural immune response, therefore, neutralizes the ability for SARS-CoV-2 to infect the body and cause COVID-19. They are therefore the most common therapeutic target for many drug companies.
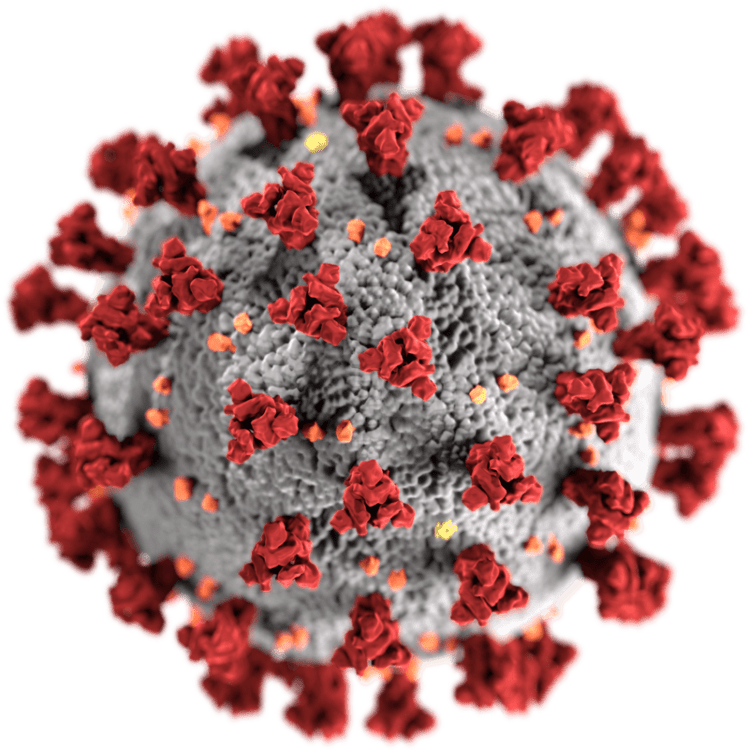
SARS-CoV-2, the virus that causes COVID-19. Spike proteins are in red.
The mRNA vaccines in development against COVID-19 carry the instructions vital for the production of spike proteins that are normally found on the surface of the novel coronavirus.
Tweet
There are a number of vaccines in development against the SARS-CoV-2 virus. Of these many vaccines, there are two mRNA vaccines that have reached Phase 3 clinical trials: Pfizer and BioNTech’s BNT162, and the Moderna mRNA vaccine.
Pfizer and BioNTech’s BNT162 vaccine completed Phase 3 clinical trials in late November. The vaccine received FDA approval via Emergency Use Authorization (EUA) on December 11, 2020 after the clinical data showed that the vaccine is safe and effective.
As a result of receiving expedited FDA approval, the Pfizer/BioNTech vaccine is currently available on the market. This makes it the fastest vaccine developed for COVID-19 — even faster than the vaccines developed as part of Operation Warp Speed. One limitation of the vaccine is that it must be stored at -70 degrees Celsius (-94 degrees Fahrenheit), though it can be stored at near-freezing temps for five days before use.
Pfizer and BioNTech’s mRNA Vaccine Is Called BNT162
As we have already mentioned, Pfizer and BioNTech’s vaccine, called BNT162, is another mRNA vaccine in development against the SARS-CoV-2 virus, which causes COVID-19. The three companies developed a version of the mRNA vaccine that would require a booster shot. This means that the vaccine would require two doses.
This vaccine, though it is called the Pfizer/BioNTech or Pfizer vaccine, was developed in collaboration with Chinese drug maker Fosun Pharma. They developed two versions of the vaccine: BNT162b1 and BNT162b2.
In May 2020, the two versions of the vaccine were developed and ready for Phase 1 clinical trials. Results of this trial showed that both versions of the vaccine elicited an immune response. Furthermore, the immune response evoked was directly proportional to the dose of the vaccine the participants received – the higher the dose, the stronger the immune response.

A vaccine must be both safe and effective. Considering that both versions of the vaccine evoke robust immune responses, it was clear that they were both effective. However, Phase 1 trial results indicate that BNT162b2 produced significantly fewer mild side effects (such as fever and fatigue) in comparison to BNT162b1. For this reason, BNT162b2 was chosen for Phase 2 and Phase 3 trials.
Now that we’ve introduced the BNT162b2 vaccine, let’s discuss how it works in more detail.
An Introduction to The BNT162b2 Vaccine
The BNT162b2 vaccine encodes a pre-fusion stabilized membrane-anchored SARS-CoV-2 full-length spike protein. “Hmm,” you might think. “That sounds much too complicated for me to understand.”
What exactly is a “pre-fusion stabilized membrane-anchored SARS-CoV-2 full-length spike protein”?
It is an entire spike protein found on the novel coronavirus molecule, which is found in a specific shape. The spike proteins are important because they allow SARS-CoV-2 to enter cells and cause a COVID-19 infection.
To understand what exactly is in the Pfizer/BioNTech vaccine, it is vital to know more about the structure of the viral spike (S) protein and how the spike protein facilitates COVID-19 infection. So, that’s what we will talk about in the next few sections.
The Structure of The SARS-CoV-2 Viral Spike Protein
By now, you’ve likely become accustomed to the iconic image of the SARS-CoV-2 virus — a round sphere with several spiky protrusions on its surface (you can also find it at the beginning of this article). The spike proteins are anchored into the membrane of the viral particle like an iceberg. The top of the spike protein sticks out, while the rest of it is anchored into the membrane. This spike protein is vital for the mediation of the novel coronavirus’s entry into host cells.
Each spike protein has two different parts, also called subunits: the S1 subunit and the S2 subunit. Both these subunits have unique functions that enable the SARS-CoV-2 virus to infect the lungs, heart, blood vessels, brain, etc. The S1 and S2 subunits are linked together at what is called the “S1-S2 subunit cleavage site.” The S1-S2 subunit cleavage site is important in terms of the mechanism of infection of SARS-CoV-2, which we will talk about next.
Mechanism of Infection for COVID-19
The novel coronavirus uses the spike proteins’ S1 subunits to bind to human cells. This binding occurs via interactions with ACE2 — a receptor found all over the body in the heart, lungs, blood vessels, brain, and so on. The S2 subunit can be thought of as a “wingman” to the S1 subunit that helps the novel coronavirus transmit its genetic information to the human cell it is infecting. The S2 subunit helps empty the viral genome from the virus into our body’s cells to cause a COVID-19 infection.
SARS-CoV-2 uses both the S1 and S2 subunits to cause a COVID-19 infection. There is an important intermediary step that we haven’t talked about yet, though, and that is a breakage that occurs between S1 and S2 subunits after the novel coronavirus’s viral RNA is transferred to a human cell via the spike protein. The S1 and S2 subunits are snapped apart from each other via certain enzymes called proteases. The exact mechanisms here are outside of the scope of this article, and it remains unclear whether the subunits separate entirely or just change shape.
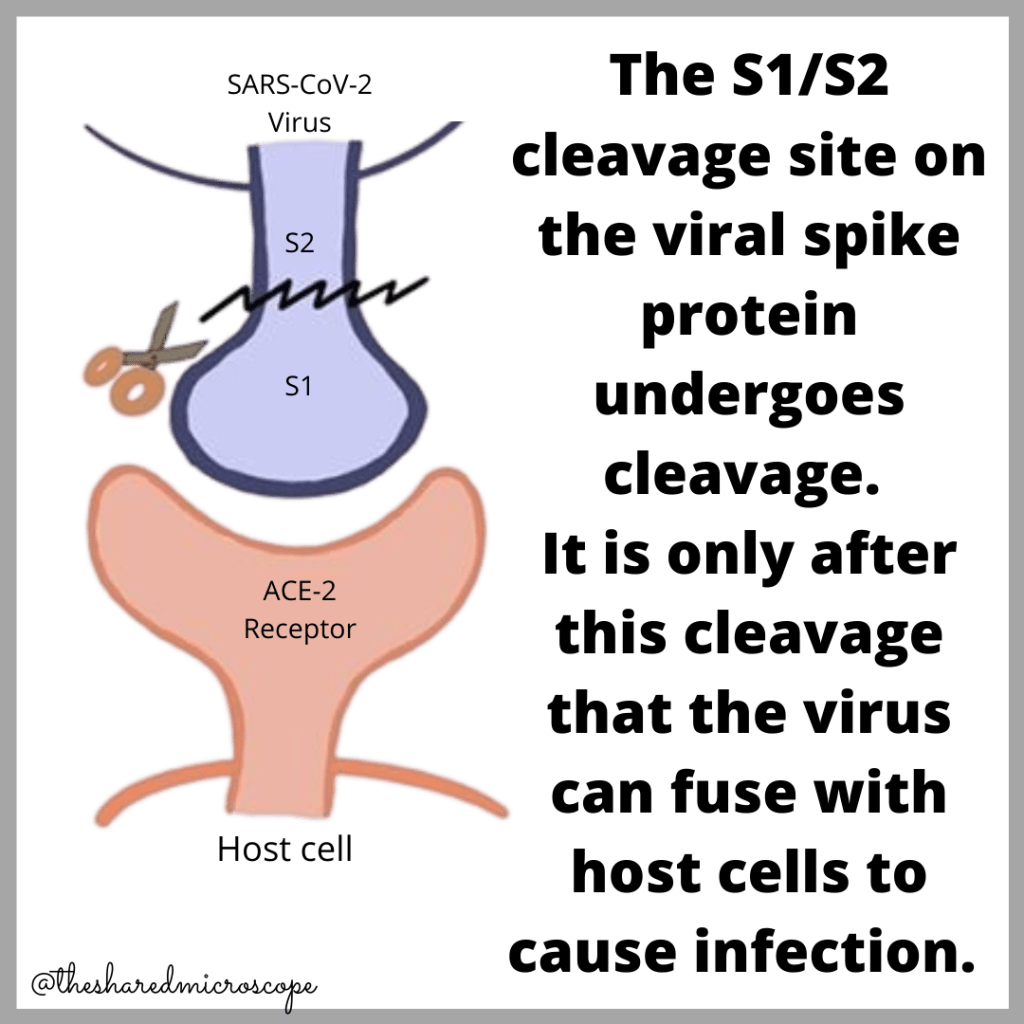
As you can imagine, after the S1 and S2 domains are cleaved or broken apart, the confirmation of the viral spike proteins change. This is somewhat like cutting a piece of paper – it will look different after you cut it. Maybe a portion of it is separated, or it’s just ripped through.
The Spike Protein Has Two Shapes: Pre-Fusion and Post-Fusion
After the SARS-CoV-2 spike protein fuses with a human cell, its shape in three dimensional space (also called its conformation) changes.
To best explain these distinct conformations, check out the following illustration by science writer and illustrator Nidhi Parekh of The Shared Microscope:
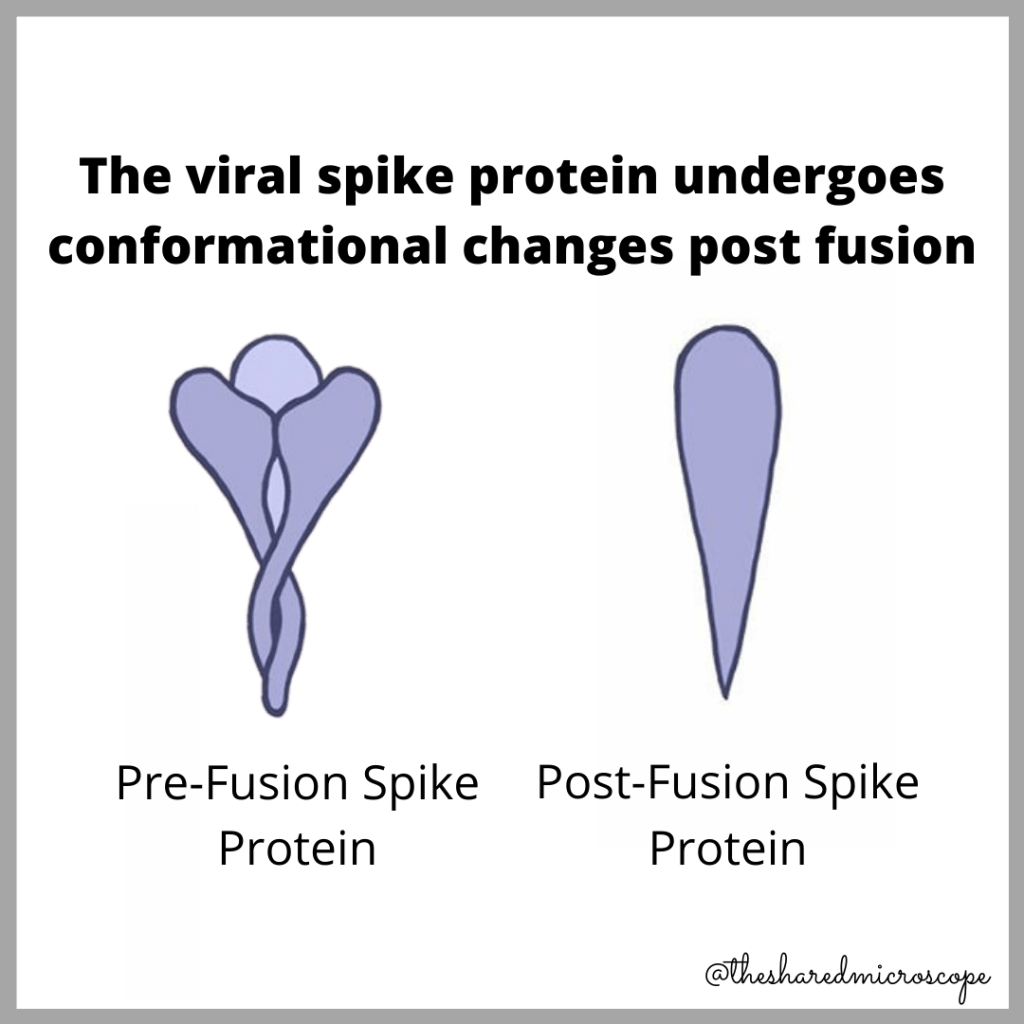
As you can see from the image above, these conformations are quite different pre- and post-fusion to the human cell. The pre-fusion conformation is how the spike protein looks before it infects our cells – when the spike protein is at the point of “starting communication” with our body’s cells. Once the virus’s spike protein connects with, and binds to, ACE2 receptors on the surface of our cells, the spike protein’s subunits change shape, perhaps to more efficiently deliver the viral genome into our cells. The fusion of spike protein to human cell causes a change in the conformation of the spike protein – from a pre-fusion state to a post-fusion state.
Once the virus fuses with one of our cells, it machinates the process of viral production to ensure that the cells are only making more copies of the virus. The result is unstoppable viral replication leading to a case of COVID-19. To prevent this coup, our cells should be able to identify the virus and stop it in its tracks before it is able to infect our cells. The vaccine teaches our body how to do this, thereby preventing infection.
What’s in the BNT162b2 Vaccine?
Why was this explanation important to understand the Pfizer/BioNTech vaccine? It’s because the BNT162b2 vaccine relies on the pre-fusion conformation of the spike protein. This is perhaps because, in an actual COVID-19 infection, our bodies will be exposed to the pre-fusion conformation of the vaccine. It is after initial “contact” with the virus that the conformation of the spike proteins changes to the post-fusion conformation. So, developing immunity to the pre-fusion conformation of the spike protein ensures that our body will be able to leverage an immune response and neutralize this exact conformation of the spike protein.
The pre-fusion spike protein is stabilized so that it cannot change its “structure.” This can be thought of as a safety feature of sorts. This vaccine also ensures that we build immunity against the spike protein, and therefore build immunity against the virus.
The Pfizer/BioNTech #COVID19 vaccine contains the mRNA needed for the human body to produce a specific form of the #SARSCOV2 spike protein that causes a COVID-19 infection. $PFE $BNTX
Tweet
In both Moderna’s and the Pfizer/BioNTech vaccines, the spike protein mRNA molecules are enclosed in a lipid nanoparticle to ensure efficient delivery into the body.
How Does the Pfizer/BioNTech Vaccine Work?
Like the Moderna vaccine, the BNT1262b2 contains instructions for our bodies to create the pre-fusion conformation of SARS-CoV-2 viral spike proteins. As we have mentioned earlier, this protein is very important to the virus’s infection mechanism – the virus cannot infect any cells in the human body without the spike protein.
The spike proteins on the surface of the virus act as a special key to the door that is our body. If we are able to make several copies of the key to study the key and understand how it destroys us, we can set up alarm systems and traps to keep the intruder (the virus) out.
This is what the vaccine aims to do – it teaches the cells in our body the shape of the “special key” so we can quickly, effectively and easily identify it in the future. Once we are aware of this key that harms us, we then respond to it by ensuring those keys are blocked using traps. These “traps” are what we call antibodies. Antibodies neutralize these “special keys” so they are unable to intrude into our cells.
If we block the intruder (the virus) before it is able to enter our cells and do any harm, we can prevent infection by the SARS-CoV-2 virus, and therefore also eliminate our risk of COVID-19 infection.
Once the mRNA vaccine BNT162b2 is administered, the mRNA instructs our body to produce copies of the “special keys” so that our body can study them. These keys are flagged up in our body as they are different, even though they are replicas – like a badly-cut key. Our immune system can then sense that something isn’t right, and will eliminate these “keys.”
With the help of the vaccine, our body is able to learn to identify these spike proteins (keys) found on the SARS-CoV-2 vaccine and make antibodies (traps) to neutralize the effect of the spike proteins. This, in turn, prevents future COVID-19 infection – evoking immunity against the virus.
The Pfizer/BioNTech Vaccine’s Current Situation
Update: This vaccine obtained FDA approval for use in adults via Emergency Use Authorization on December 11, 2020, making it the first commercially available COVID-19 vaccine. On May 10, 2021, the vaccine became authorized for emergency use in adolescents aged 12-15.
The vaccine in development by Pfizer, in collaboration with BioNTech and Fosum Pharma, has recently been in the spotlight almost on a daily basis. The Chief Executive of Pfizer has repeatedly said he hopes to answer questions on the safety and efficacy of the vaccine as early as October 2020.
In July, the companies launched Phase 2/3 trials with 30,000 volunteers in the US, Argentina, Brazil and Germany. The world is still awaiting these results. However, interim results suggest that volunteers see ‘mostly mild to moderate’ symptoms (like fatigue and headache) on receiving the vaccine – which is very promising. Earlier this month, the companies announced that they are seeking to expand their trial in the U.S. to 44,000 patients.
Will this vaccine be our dark horse? Our knight in shining armor? Will it bring humanity a step closer to normal? There are a number of unanswered questions on all of the vaccines, but if this vaccine is successful, the two companies are set to manufacture 1.3 billion doses of their COVID-19 vaccine by the end of 2021.
Nidhi Parekh is a science writer and illustrator blogging at The Shared Microscope. Check them out on Twitter, hop over to their Facebook, or visit their Instagram for informative, effective illustrations of concepts in biology.
13 thoughts on “Pfizer and BioNTech’s mRNA Vaccine for COVID-19: How Does It Work?”