Updated October 30, 2020
By Nidhi Parekh, The Shared Microscope
Read on to learn about the origins of the Oxford vaccine, how it works, and why it is expected to be so promising in the global efforts to end the COVID-19 pandemic. Lea este artículo en español.
Check out other posts in our COVID-19 vaccine series:
[Moderna | Oxford/AstraZeneca | Pfizer/BioNTech | Johnson & Johnson | Novavax | Sinovac]
Table of Contents:
- How Does the Vaccine Development Process Work?
- Better Together: Oxford University and AstraZeneca Team Up to Accelerate COVID-19 Vaccine Development
- How does the Oxford University/AstraZeneca vaccine work?
- Training Our Immune System to Defeat COVID-19
- What Is a Viral Vector?
- Pasting Together Viruses to Make Vaccines
- So What Is the ChAdOx1-nCov19 Vaccine?
- What Are the Advantages of a Viral Vector Vaccine?
- What Evidence Is There to Support the ChAdOx1-nCov19 Vaccine?
- Clinical Trials for the Oxford/AstraZeneca Vaccine
How Does the Vaccine Development Process Work?
Before a vaccine can be released onto the commercial market, it must undergo rigorous testing in clinical trials. The purpose of the clinical trials process is to ensure that the COVID-19 vaccine is both safe and effective. The COVID-19 vaccine development process would normally take years to complete. However, due to the urgent need for COVID-19 vaccines worldwide, there is an international effort to expedite vaccine development while still ensuring safety and efficacy to the greatest extent possible.
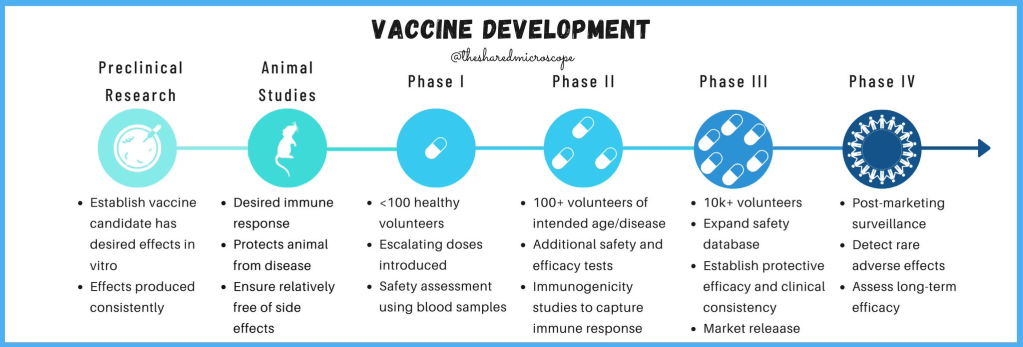
Vaccine development timeline, from preclinical laboratory research to clinical trials with human subjects. Illustration by The Shared Microscope.
Better Together: Oxford University and AstraZeneca Team Up to Accelerate COVID-19 Vaccine Development
Just weeks after the SARS-CoV-2 virus was detected in Wuhan, China in December 2019, a team of Oxford scientists led by Dr. Sarah Gilbert got to work on designing a vaccine. On the 10th of January 2020, just weeks after the SARS-CoV-2 virus was detected in Wuhan, a team of researchers began their efforts to develop a vaccine. Since then, the Oxford team has identified a vaccine candidate, and completed Phase I trials. With the help of AstraZeneca, the vaccine has now entered Phase II/III trials in countries like Brazil and South Africa. AstraZeneca has partnered with the Oxford University team to accelerate development and large-scale vaccine production. Remember, for a vaccine to be developed and released to the public for use, it must pass all the necessary hurdles. This is to ensure safety and efficacy of the vaccine for use in large numbers.
You can read about what it’s like to participate in the clinical trials for the Oxford/AstraZeneca vaccine here.
How does the Oxford University/AstraZeneca vaccine work?
Curious about how vaccines work in general? Check out this post from The Shared Microscope on Instagram.
The Oxford/AstraZeneca vaccine uses a non-replicating viral vector to introduce vital information about a pathogen to the body of the host. But what information are we introducing, and what even is a viral vector? This is a bafflingly complex topic and to aid your understanding, we have simplified each part of it below.
Training Our Immune System to Defeat COVID-19
Like most of the vaccines being made against the novel coronavirus, the Oxford University vaccine, too, exploits the spike protein of the SARS-CoV-2 virus. The spike protein, as discussed in our earlier post, plays a very important role in the infection process. Without the spike protein, the SARS-CoV-2 virus cannot enter the host cell, where it typically makes more copies of itself. In other words, the spike protein is important for the virus’s ability to facilitate a COVID-19 infection. Neutralizing the effect of the spike protein, therefore, neutralizes the ability of the novel coronavirus to cause infection. The SARS-CoV-2 spike proteins are therefore a very important and potent therapeutic target. The Oxford/AstraZeneca vaccine trains our bodies to defend against the SARS-CoV-2 virus and prevent COVID-19 infection.
The Oxford/AstraZeneca vaccine trains our bodies to defend against the SARS-CoV-2 #virus and prevent COVID-19 infection. — @TheSharedScope #covid19 #scicomm #sciencetwitter
Tweet
What Is a Viral Vector?
In biology, a viral vector is a tool used by scientists to introduce genetic material into cells.
Viral vectors provide a convenient means to deliver antigens to the body to help us develop immunity to them. The antigens used in viral vector vaccines are weakened forms of bacteria or viruses. Once the vector introduces the body to an antigen, our body then develops immunity to the antigen.
A viral vector can be the basis of a vaccine to develop immunity to a virus — in fact, it is the driving concept behind the Oxford/AstraZeneca COVID-19 vaccine. Used in this way, a viral vector can be a modified version of a virus that is not only incapable of causing disease, but, when introduced into the body, can help us develop immunity to the virus.
One analogy that can be used to explain viral vectors is that of “cut and paste.” We are all familiar with “cut and paste” — one might cut pictures out of a magazine and glue them together to produce decoupage art, or we may cut and paste text when we are working on our computers.
We can think of viral vectors as formed by selectively cutting out certain genes which perform different, crucial functions, making various copies of these functionally relevant genes — and adding them together to other genes which add new functions. In this way, viral vectors can be used to change how a virus works — to make a virus less infectious, for example. In biology, this cutting and pasting is called recombination. Different parts of viruses are combined together in new ways to create what is called a “recombinant” virus.
Viral vectors, such as those used in the Oxford/Astrazeneca #COVID19 vaccine, are created via cut-and-paste — or what scientists call recombination. @TheSharedScope #biology #scicomm #sarscov2 #vaccine.
Tweet
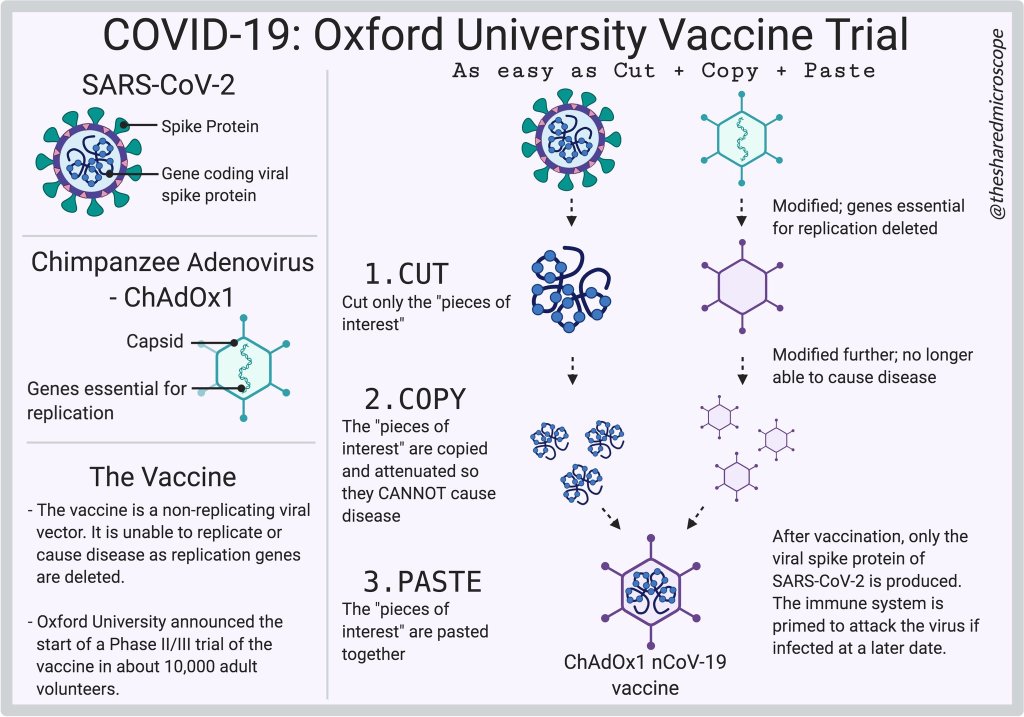
The Oxford/AstraZeneca Vaccine for COVID-19 uses a “Cut, Copy, Paste” Approach. Illustration by The Shared Microscope.
Just take a virus, take out the parts that cause infection, add parts from another virus that do not cause infection (but do help you build immunity to that other virus), and voilà — you’ve got a viral vector which has some properties of both viruses, but is neither of those viruses. And to reiterate, cannot cause disease.
Pasting Together Viruses to Make Vaccines
As we discussed in the previous section, cutting and pasting of different viruses is called recombination. The Oxford/AstraZeneca COVID-19 vaccine is a recombinant virus vaccine made of two different viruses. One of the viruses is SARS-CoV-2, the virus that causes COVID-19. The other virus in the recombinant vaccine is a chimpanzee adenovirus called ChAdOx1. Don’t worry if you don’t know anything about ChAdOx1 — we will discuss it more in this section.
Normally, viruses are infectious, and the novel coronavirus is no exception. But with recombination, infectious viruses can be cut-and-pasted so that they are no longer infectious — the genetic material that is responsible for the virus’ infectious nature are simply cut out. Recombination allows scientists to create a non-infectious virus with characteristics that they desire from cutting-and-pasting different viruses together, as has been done with the Oxford/AstraZeneca vaccine.
In the recombinant viral vaccine developed by Oxford and AstraZeneca, the relevant parts of the two viruses are pasted together. This recombination is a frontrunner in the race to develop a COVID-19 vaccine.
In the vaccine developed by Oxford and AstraZeneca, the relevant parts of the two viruses are pasted together. The vaccine is a frontrunner in the race to end the COVID-19 pandemic. — @TheSharedScope #COVID19 #oxford #astrazeneca
Tweet
Adenoviruses Are Commonly Used in Viral Vectors
One type of virus that is often used as a base for viral vectors are adenoviruses. Adenoviruses commonly infect people and cause mild symptoms similar to a cold or flu. This type of virus is also the basis for the viral vector used in the Oxford COVID-19 vaccine.
Although there are various kinds of adenoviruses, the Oxford University vaccine uses the Chimpanzee Adenovirus (ChAdOx1) as its base. Then, into this virus, the genetic material that encodes for the SARS-CoV-2 spike protein is added. The result is a virus that is not infectious but can cause the body to develop immunity to the spike protein and help stop the SARS-CoV-2 virus from entering and infecting cells.
The Oxford/AstraZeneca COVID-19 Vaccine Uses a Chimpanzee Adenovirus Called ChAdOx1
The basis for the Oxford/AstraZeneca COVID-19 vaccine is a chimpanzee adenovirus called ChAdOx1. Why did the Oxford researchers choose a chimpanzee adenovirus? The reason for this is simple. Human adenoviruses cause the common cold. These are viruses most of us will have some immunity to, as most of us are infected by these almost every year. So, if we were to use a human adenovirus as a viral vector, our immune system would destroy the human adenovirus as soon as it is detected by our body. The vector would therefore not work as effectively (if at all), as it would be destroyed before it could provide the host body (the human) with instructions on how to fight the novel coronavirus, which causes COVID-19.
Since humans aren’t exposed to the Chimpanzee Adenovirus (ChAdOx1) very often, its introduction would elicit an immune response in our cells. Inside this ChAdOx1 vector is the genetic information of only the SARS-CoV-2 spike protein, cut-and-pasted. The introduction of the ChAdOx1 would elicit an immune response in our cells. To ensure that the ChAdOx1 cannot take hold of our system and make thousands of copies of itself, it is important that genes vital for its replication are “cut” out — deleted. What happens here, therefore, is that the virus will enter the human cell (singular) and infect it, but the disease is no longer able to spread. Each viral vector introduced to the body can only infect one cell, as opposed to infecting one cell and replicating to infect more.
With this ChAdOx1 vector (with the spike protein code cut-and-pasted inside), it would simply be the case that our bodies recognize the chimpanzee adenovirus as foreign, and then move to destroy it, and, in the process, become aware of the structure of the SARS-CoV-2 spike protein. As explained in our previous post, this is important because our body can then learn to identify these spike proteins, and then produce antibodies against them. This has the effect of neutralizing SARS-CoV-2, because it prevents the virus from infecting cells and causing COVID-19. SARS-CoV-2 spike proteins are a very potent therapeutic target, and is being exploited in the production of a number of vaccines against COVID-19.
In normal infection scenarios, all of the genes essential for replication of the virus are intact i.e. they are not being deleted from the virus. In this case, the viruses are able to infect the cell, take over its machinery, and replicate by the dozen. These would then spread to other cells in the body and infect them too. It is when the viruses replicate and infect a number of other cells, that the disease is said to have manifested. More and more cells will therefore be infected. A good strategy is therefore to prevent it, by deleting the genes vital for replication of the vector virus.
So What Is the ChAdOx1-nCov19 Vaccine?
The ChAdOx1-nCov19 vaccine, developed by Oxford/AstraZeneca, is in essence a cut-and-paste version of both the chimpanzee adenovirus (ChAdOx1) and the SARS-CoV-2 virus. This vaccine uses the ChAdOx1 as a viral vector, and inserted into that, is the genetic information for the SARS-CoV-2 virus spike protein. For reiteration, the ChAdOx1 is unable to replicate, as some genes essential for replication have been deleted. So essentially, the Oxford/AstraZeneca vaccine looks like a ChAdOx1 virus on the outside, but its contents are that of the SARS-CoV-2 spike protein.
Wondering how the candidate #COVID19 vaccines work? The Oxford/AstraZeneca #vaccine helps us develop immunity to a crucial part of SARS-CoV-2 — the spike protein. $AZN @TheSharedScope #scicomm
Tweet
The genetic information to code for the spike protein is added to the ChAdOx1 vector. The viral vector (ChAdOx1) acts like a capsule inside which it carries its most prized possession — genetic information to produce the SARS-CoV-2 spike protein. The non-replicating vector will infect a single human cell, where it produces the antigen we want — the SARS-CoV-2 spike protein — to elicit an immune response.
Each viral vector that is introduced to the body can therefore only infect ONE cell. The adenovirus vector infection is unable to spread to all the cells in our body, because it cannot replicate. So, we do not become infected with the recombinant virus, and our body, in theory, learns to defend against both viruses — SARS-CoV-2, which causes COVID-19, and ChAdOx1. That way, it is thought, our bodies learn to resist COVID-19 infection.
Our body learns to defend against both viruses — SARS-CoV-2 which causes #COVID19, as well as ChAdOx1 — included in the recombinant virus vaccine developed by Oxford and AstraZeneca. @TheSharedScope
Tweet
What Are the Advantages of a Viral Vector Vaccine?
A viral vector has a number of features that are particularly attractive for vaccine use. These advantages of viral vector vaccines include, but are not limited to:
- The ability to grow/produce the vaccine in a lab
- The inability of the vaccine to integrate into host genes
- Their stability
- Quick to make as compared to conventional vaccines (which can take on average 5-10 years), since it is based on part of the virus rather than the entire virus (more on this in our previous post)
- The ability to manufacture them in large scale under very controlled conditions
- Their inability to replicate and spread through the body, which means they can also be used in individuals who are immunocompromised
- Their ability to produce a very strong immune response – which means another “booster” dose may not be required
- Induction of both humoral and cell-mediated immune responses
What Evidence Is There to Support the ChAdOx1-nCov19 Vaccine?
The Oxford Group has previously used the ChAdOx1 viral vector to protect against another coronavirus – the Middle East Respiratory Syndrome or MERS. SARS-CoV-2 has structural similarities with MERS. A similar type of spike protein is found on the MERS coronavirus (MERS-CoV), just as it is with SARS-CoV-2.
The MERS-CoV was initially a virus that causes cold-like symptoms in camels, but it jumped species and, because of the similarity of the entry site of cells in camels and humans, the MERS-CoV started to infect humans.
The gene that encodes the spike protein in MERS-CoV was then put into the ChAdOx1 vector and testing began at Oxford. Preclinical testing was carried out in animal models and Phase 1 clinical trials were also performed. The results of the animal studies have recently been published. The results of the Phase 1 clinical trials in humans were also published just yesterday.
The results of the Oxford studies showed that the antibodies against the MERS-CoV spike protein were higher in subjects injected with the ChAdOx1-MERS vaccine even a year after they were vaccinated. The immune response showed that the vaccine protected individuals and animal models from disease using both a cellular and humoral response (i.e. results showed an increase in T cells, and neutralizing antibodies). For more on antibodies, please see our previous post. Based on this previous research done using MERS-CoV, it was possible to quickly discern what doses to use for the SARS-CoV-2 clinical trials.
Clinical Trials for the Oxford/AstraZeneca Vaccine
It took just a few months for the Oxford/AstraZeneca COVID-19 vaccine to go from preclinical trials in the laboratory on January 10, 2020, to human clinical trials on April 23, 2020.
It took just a few months for the Oxford/AstraZeneca #COVID19 #vaccine to go from the laboratory to clinical trials. — @TheSharedScope #scicomm #sciart #sciencetwitter
Tweet
For the ChAdOx1-nCov19 vaccine, preclinical studies were carried out on mice. The vaccine has the “nCoV19” as research began on this before the novel coronavirus was officially named SARS-CoV-2. In the study, mice were immunized using the ChAdOx1-nCov19 vaccine. Results showed that neutralizing antibodies were produced in the mice, protecting them from infection when exposed to the SARS-CoV-2 virus.
Phase I trials of ChAdOx1-nCov19 vaccine started on April 23, 2020. 561 young and healthy individuals were vaccinated with the ChAdOx1-nCov19 vaccine, out of which 10 were vaccinated with 2 doses of the ChAdOx1-nCov19 vaccine. Another 551 were recruited for the control wing of the experiment, and were given licenced vaccines against meningitis. Since then, the Phase II/III trials also began. Here, the age range of people on which studies were carried out was expanded to include older people and children, so as to further study efficacy and safety (for more on vaccine development, feel free to visit our previous post). The study is still ongoing and therefore the results are currently unknown.
The vaccine has since been licensed to AstraZeneca, who will help accelerate development and large-scale production. AstraZeneca has committed to supply Europe 400 million doses of the vaccine, using manufacturers from different countries. They have also said they will not be taking any profits during the pandemic, and will support the ongoing clinical trials in Brazil and South Africa. AstraZeneca have also started manufacturing at risk, without knowing whether the vaccine is effective against SARS-CoV-2 in humans. The World Health Organization (WHO) believes that this vaccine is the contender in the race to find the COVID-19 vaccine, and end this pandemic.
For more on the Oxford/AstraZeneca vaccine, please check out this YouTube video:
This post was written by Nidhi Parekh of The Shared Microscope. Check out The Shared Microscope on Twitter, hop over to their Facebook, or visit their Instagram for more fun and informative illustrations of biology concepts.
26 thoughts on “How Does the Oxford University/AstraZeneca Vaccine for COVID-19 Work?”